Director of Inorganic Chemistry Laboratory: C. Dendrinou-Samara, (Professor)
Secretary: Ch. Karapetsa
Inorganic Chemistry Laboratory was established in 1939 and is among the oldest one in the Chemistry Department.
Undergraduate level: Teaching and lab practices in the field of Inorganic Chemistry and General Chemistry for students of the Department of Chemistry, as well as students of other Departments (Physics, Pharmacy and Geology). Elective courses related to Nanochemistry, Materials Chemistry, Bioinorganic Chemistry, Natural Methods in Inorganic Chemistry, Organometallic Chemistry and Catalysis, Teaching of Chemistry, Radical Chemistry and Nuclear Chemistry. Research dissertations.
Postgraduate level: Inorganic Chemistry Laboratory participates at the Postgraduate Program “Synthetic Chemistry, Biochemistry and Applications” and trains postgraduate students for a Master’s Degree and PhD Diploma.
Facilities for research and educational purposes: X-ray diffractometer (crystal structure analysis), Spectrophotometers (infrared IR, ultraviolet visible UV-vis, fluorescence), Electrochemical device of circular voltammetry, Thermal analysis, Magnetic measurements at room temperature, Viscometer, α- and γ-Spectroscopy (NaI and Ge detectors), X-ray fluorescence.
Coordination Compounds
Synthesis, physicochemical characterization and structure determination by single-crystal X-ray diffraction.
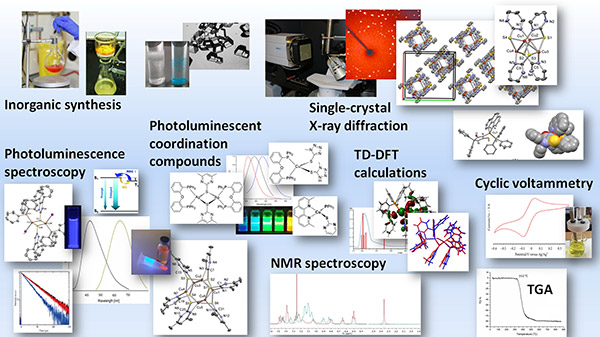
Biological/Pharmaceutical Interest Coordination Compounds
Synthesis and relationship between structure and bioactivity of complexes (DNA and albumin interaction, antioxidant, antimicrobial, anticancer, anticholinergic activity)
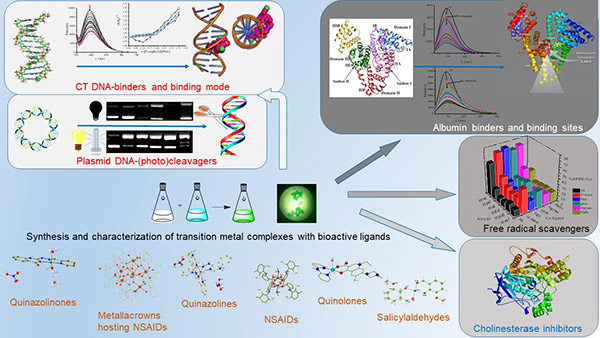
Technological Interest Coordination Compounds
Synthesis, photophysical and magnetic properties. OLED emitters, photosensitizers and catalysts for hydrogen production, photosensitizers for dye-sensitized solar cells, single molecule magnets.
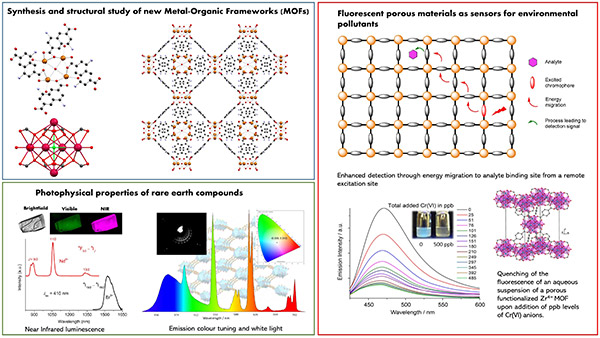
Nanomaterials
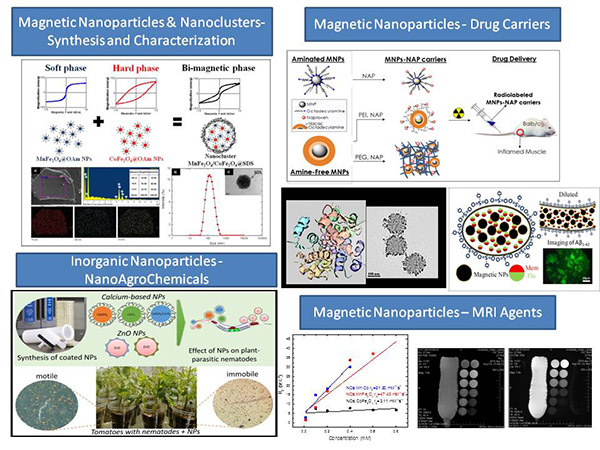
Inorganic Nanoparticles: Synthesis, characterization, biomedical activity (interaction with DNA, antimicrobial, antioxidant, anticancer activity).
Magnetic nanoparticles: Synthesis, characterization, biomedical/theranostic applications such as diagnostic techniques (MRI agents), therapy (hyperthermia) and drug delivery.
Radiochemistry-Nuclear chemistry
Environmental radiochemistry: Adsorption of radionuclides on biomass, zeolites and synthetic materials;Determination of natural radioactivity in environmental samples (soils, sediments, water) by γ-spectroscopy; Separation and determination of radionuclides by α-spectroscopy.
Application of nuclear techniques in materials research: neutron activation analysis, X-ray fluorescence and nuclear reaction analysis.
Crystallography
Single-crystal X-ray diffraction using the X-ray diffractometer of our laboratory. The method fully clarifies and illustrates the molecular and crystalline structure of the test compound. The term molecular structure means the stereochemical arrangement of the atoms forming the test compound. The term crystal structure describes the arrangement of all the contents in the crystal such as hydrogen bonds, van der Waals and π-π type interactions with the final result being the explanation and prediction of properties of the compound in solid state.

| NAME | ACADEMIC RANK | TELEPHONE | |

|
Akrivos Pericles | Professor | 231-099-7706 |

|
Dendrinou – Samara Catherine | Professor | 231-099-7876 |

|
Noli Fotini | Professor | 231-099-7997 |

|
Hatzidimitriou Antonios | Αssociate professor | 231-099-7748 |

|
Lazarides Theodoros | Αssociate professor | 231-099-7853 |

|
Psomas George | Αssociate professor | 231-099-7790 |

|
Angaridis Panagiotis | Assistant Professor | 231-099-7437 |
|
|
Karagiannidis Petros | Professor Emeritus | |

|
Manousakis Georgios † | Professor Emeritus | |
|
|
Tossidis Ioannis | Professor Emeritus | |
|
|
Tsiamis Chris | Professor Emeritus | |

|
Karapetsa Christina | Specialized Technical Laboratory Staff | 231-099-7747 |

