
Nikolakaki Eleni
Professor
Biochemistry LaboratoryAristotle University of Thessaloniki
Faculty of Sciences,
School of Chemistry
54124, Thessaloniki
Our main objective is the elucidation of signaling pathways in eukaryotic cells. More specifically, we focus on the SR family of protein kinases (SRPKs), which phosphorylate serines within arginine/serine (RS) dipeptide motifs. Most of our knowledge regarding SRPKs comes from studies on SRPK1, which was the first member of the SRPK family to be purified and characterized.
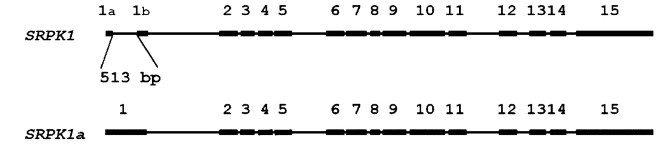
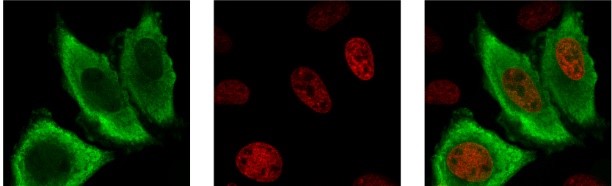
We studied in the past the expression of SRPK1 in normal and cancer cells. In addition, we cloned and characterized a new SRPK1 isoform, SRPK1a, which contains an insertion of 171 amino acids at its N-terminal domain and is similar to SRPK1 in substrate specificity and subcellular localization. The level of expression of SRPK1a is low, but this isoform is far more active than SRPK1. Even more interestingly, an increase in the SRPK1a/SRPK1 ratio leads to differentiation of K562 erythroleukemia cells. Using mass spectroscopy analysis we have found a number of proteins that interact specifically with the extra segment of SRPK1a and we are currently trying to probe into their biological function.
We followed a mutagenesis approach to probe into the structure, function and regulation of the subcellular localization of SRPK1. In this respect we have shown that phosphorylation of serine 51 by casein kinase 2 (CK2) increases kinase activity, while mutations of a number of cysteines within the spacer domain, that separates the two catalytic subunits, result in loss of activity and retention of the kinase in the cytoplasm. In parallel, we studied in detail the phosphorylation mechanism employed by SRPK1, using as a substrates various proteins that contain RS dipeptides, such as LBR, acinus and PGC-1a (Peroxisome proliferator-activated receptor Gamma Coactivator-1 alpha). We propose that the tendency to self-associate and form aggregates is a general property of RS domain-containing proteins and could be attributed to their disordered structure. RNA binding or SRPK1-mediated phosphorylation prevents aggregation and may serve to modulate the RS domain interaction.
Recent data suggest that SRPK1 plays a pivotal role in cancer. In this respect our current studies focus on the involvement of SRPK1 in proliferation and apoptosis of cancer cells. Furthermore, we are trying to develop novel and potent inhibitors targeting specifically SRPK1.

