
Dr. Evgenios Kokkinos has a degree in Chemistry from Aristotle University of Thessaloniki, majoring in Chemical and Environmental Technology (MSc), and currently he is a Postdoctoral researcher in the same department. His doctoral research was done at the Chemical Engineering Department of the Aristotle University of Thessaloniki. His main research interests are the removal of toxic metals from water and flue gases, as well as their recovery from industrial waste, in the concepts of good housekeeping and circular economy.
Research
Removal of toxic metals from aqueous and gas streams
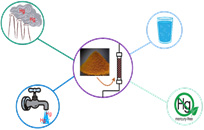
The main technology applied in the field for toxic metals’ removal from liquid and gaseous streams is adsorption. Its main advantage is the ability to achieve residual concentrations of pollutants below the maximum permitted emission and consumption limits, which of course have been institutionalized at extremely low levels due to their hazardous effects to humans and the environment. The removal of toxic metals, such as Hg, MeHg, As, Sb, Ni, Cd, etc., in the Laboratory of Chemical and Environmental Technology is carried out by synthesizing inorganic (binary iron-manganese nanomaterials) and organic (activated carbon) adsorbents.
Cr(III) recovery from tanning waste and its reuse
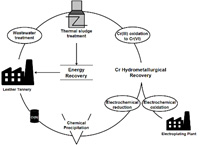
Tanning of the skin is done chemically, using Cr(III) salts, so that the final product acquires the desired properties. The tanning reactions present low efficiency and, as a result, 40% of Cr(III) ends up in the corresponding waste. In the context of circular economy, but also the good housekeeping of waste, in the Laboratory of Chemical and Environmental Technology was achieved an over 90% Cr(III) recovery by applying the principles of hydrometallurgy and the ability to be fed back to the tannery. In addition, energy recovery and its electrochemical oxidation to Cr(VI) were investigated, aiming to promote it in other industries, e.g. electroplating.

